Neurotech study finds children with autism benefit from cannabis product
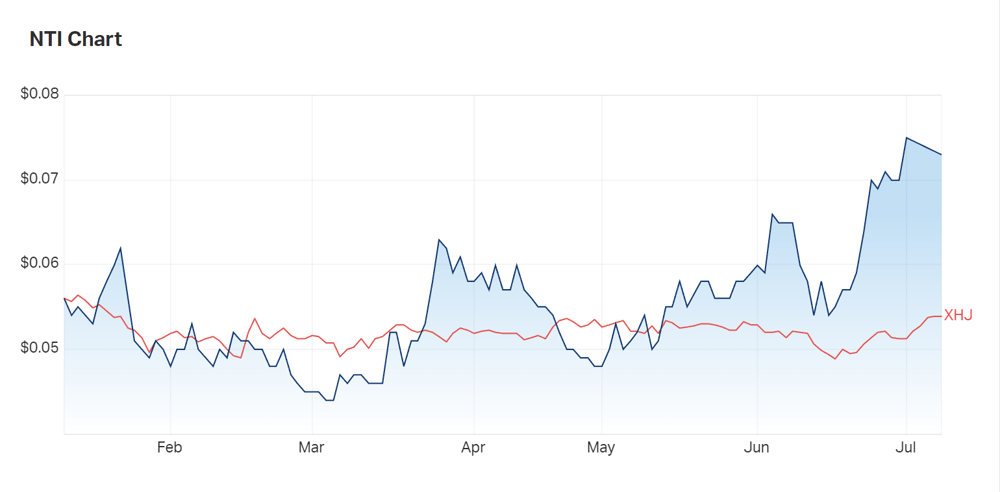
Neurotech (ASX:NTI) is up over 25% YTD and outperforming the ASX200 - is it onto a big thing?

Source: Unsplash
Mentioned
KEY POINTS
- Neurotech’s low-THC (<0.3%) NTI164 strain, taken orally, reportedly assists with reducing symptoms of Autism Spectrum Disorder (ASD)
- Participant ages ranged between 8 to 17
- Study found dosages between 5mg/kg to 20mg/kg safe with no impact on vital signs or weight
Neurotech International (ASX:NTI) has today announced its patent NTI164 product has been proven to reduce ASD symptoms in children and teenagers.
The low-THC content cannabinoid saw 64% of patients with ASD reporting “much improved” symptomology after a trial using the oral drugs.
A majority of patients reported “Decided Improvement” of symptoms, and a small number reported an effective complete remission of symptoms.
Neurotech continues to engage with the US FDA to acquire Investigational New Drug (IND) status for NTI164, which would see it fast-tracked through preliminary approval stages.
Company hopes to replace Risperidone
Neurotech notes the only FDA approved drug currently given to children with ASD is Risperidone, an antipsychotic with undesirable side effects.
“We are encouraged by the efficacy shown by NTI164 in this trial and we are looking forward to the planned initiation of a Phase II/III trial to further assess,” Chief Investigator at Monash Children’s Hospital Professor Fahey said.
Neurotech chairman Brian Leedman offered more enthusiasm.
“We cannot underestimate the significance of the results from our landmark trial…NTI is now a step closer in the development timeline to introducing a treatment option for paediatric ASD,” Leedman said.
“The fact results are based on a 28 day trial, the company is very excited that it is genuinely opening up a new treatment pathway.”
The company’s excitement may be better understood when looking at forecasts for the value of the ASD treatment market.
Recent research shows the sector is expected to be worth over US$5bn by 2028.
What are investors thinking about Neurotech?
While the share price has dipped its toes into the red today, investor interest in Neurotech is clear.
Since July last year, the company’s share price has risen 30.3%.
Over 20% of that growth has occurred since the start of 2022.
Versus the ASX200 index on a one year basis, Neurotech’s share price performance is up by 38.9%.
Neurotech has a market cap of $50.9m.