PharmAust patients to continue trial using…livestock wormicide
The drug Monepantel has a desirable side-effect in humans which could potentially treat MND

Source: iStock
Mentioned
KEY POINTS
- PharmAust is running a six-person trial using monepantel (MPL) to treat Motor Neurone Disease (MND)
- The company also continues to use the drug in a Covid-related trial in Europe
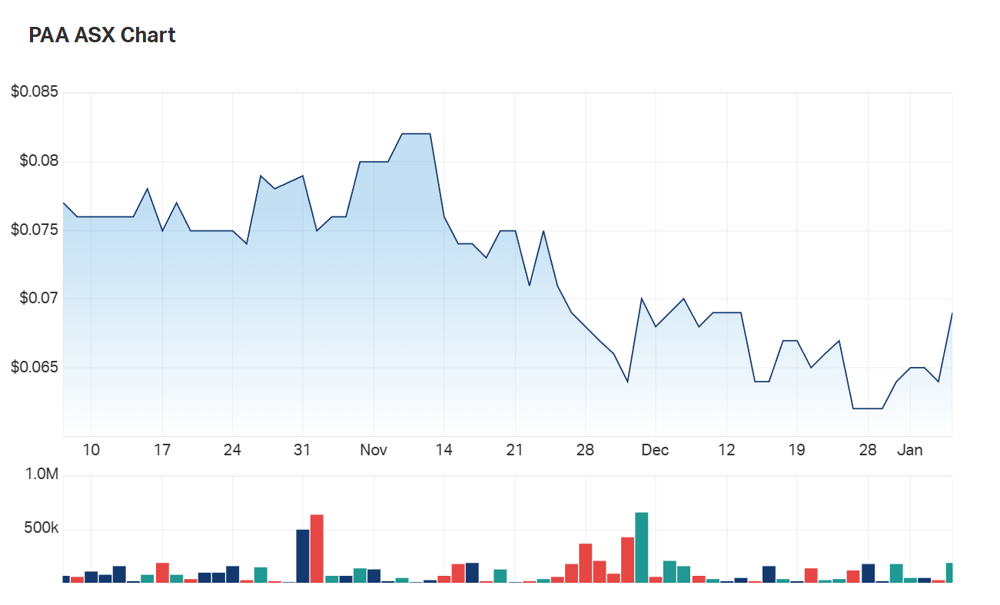
- Shares were up 7% in the second hour of Friday trade
PharmAust Limited (ASX:PAA) shares are rallying this morning after the company revealed the first batch of patients using Monepantel (MPL) in a trial to treat MND have elected to persist with the trial.
This is significant news for the technical team at PharmAust.
The company is using MPL not for its intended purpose—a wormicide for livestock—but for its off-target side effects.
Wanted side effects
“Monepantel shows off-target activity, inhibiting a cellular signaling system controlled by [enzymes]. This stops cancer growth and reduces protein accumulation in diseased cells,” the authors of a paper sponsored by PharmAust wrote in May 2021.
In May last year, the company kicked off a Covid-related trial testing the effects of the drug in unvaccinated patients.
A lack of participants in Australia forced it to recruit in Eastern Europe.
Patients happy to keep using MPL
Now, nine months later, six participants in the MND trial investigating MPL report no bad side effects and a willingness to continue participating.
Whilst using a veterinary medicine to treat Covid would be on the nose for risk-off investors, this outcome will undoubtedly allay some concerns - which is being reflected in the share price this morning.
Patients will now take increasing doses of MPL as the trial moves to its next stage. A new round of recruitment will be launched to expand the sample size.
While the company does not today report any results of the study, the news will be of interest to healthcare investors.
Should the PharmAust MND trial turn up desirable outcomes (and that’s a while off yet), the company seeks to list MPL for MND under orphan drug status on the Federal Drug Administration’s (FDA) books in the USA.