Mayne enters US women’s health market with NASDAQ-listed partner for $205m; shareholders spooked
Deal with NASDAQ heavyweight gives Mayne great exposure to US, but obligations onerous

Source: iStock
Mentioned
KEY POINTS
- Mayne Pharma has signed a licence agreement with NASDAQ-listed TherapeuticsMD to sell five women’s health products in US
- Deal sees Mayne paying TherapeuticsMD A$205.5m; further performance payments due to TherapeuticsMD
- Products’ revenues in Q3 CY2022 stand at A$30.6m revenue; gross profit A$25.1m
In a deal the company expects to be immediately material to its own revenues and profits, Australia’s Mayne Pharma (ASX:MYX) is entering the US women’s healthcare product market with NASDAQ-listed partner TherapeuticsMD.
Executing a licencing agreement with Therapeutics, Mayne will hand the former $205.5m to fund the product-line acquisition.
Payments to Therapeutics MD by Mayne will be made on sales-based milestones; for every US$100m in revenue made each year, Mayne will need to hand TherapeuticsMD $5m.
Mayne will also pay royalties of US$3m a year for 12 years.
Onerous obligations scare shareholders
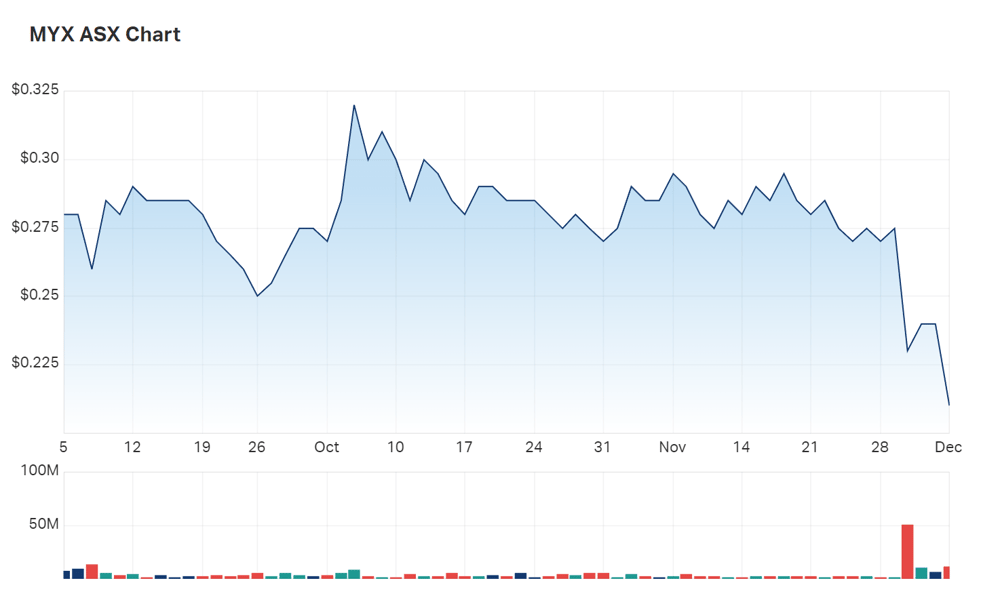
Perhaps unsurprisingly, some shareholders have seen it fit to scatter. Maybe Pharma shares are down -8% in the early second hour of trade.
The US$140m acquisition will be funded in part “through a binding commitment from Rubric Capital Management for approximately US$27.95m…the maximum number of shares issued to Rubric will be the lesser of 178.8m shares and 9.61% of Mayne Pharma’s issued capital.”
Mayne will now sell five products already on the shelves in the US with the company expecting material impact on revenue and profits to be immediate.
Across the five products Mayne today acquires a licence for, Q3 CY2022 profits from those items alone stood at $25.1m. The currently weaker Australian dollar against the greenback is also a strong point of today’s deal.
What will Mayne Pharma be selling?
Mayne Pharma picks up the licence for the following products:
BIJUVA - A menopause treatment to treat hot flushes (licence until 2032)
IMVEXXY - Another menopause treatment used to treat vaginal pain (licence until 2034)
ANNOVERA - A long-term self-insertable contraceptive product (licence until 2039)
BocaGreenMD - Prenatal vitamins emphasising natural ingredients
VitaMedMD - Prenatal vitamins
Mayne expects the deal to close by the end of the calendar year.