Actinogen up 30% on strong Alzheimer’s trial results, says Xanamem product can slow progression
Company now moving ahead to more clinical trials; today's data also supports diagnostic breakthrough
Source: Unsplash
Mentioned
KEY POINTS
- Actinogen says phase 2 trial data for its Xanamem oral medication product shows ability to slow progression of Alzheimer’s Disease (AD) in early onset patients
- Company also states its ability to diagnose early AD by screening blood tests
- Actinogen using FDA-approved measurements in its trial boosts chance of painfree progression towards approval
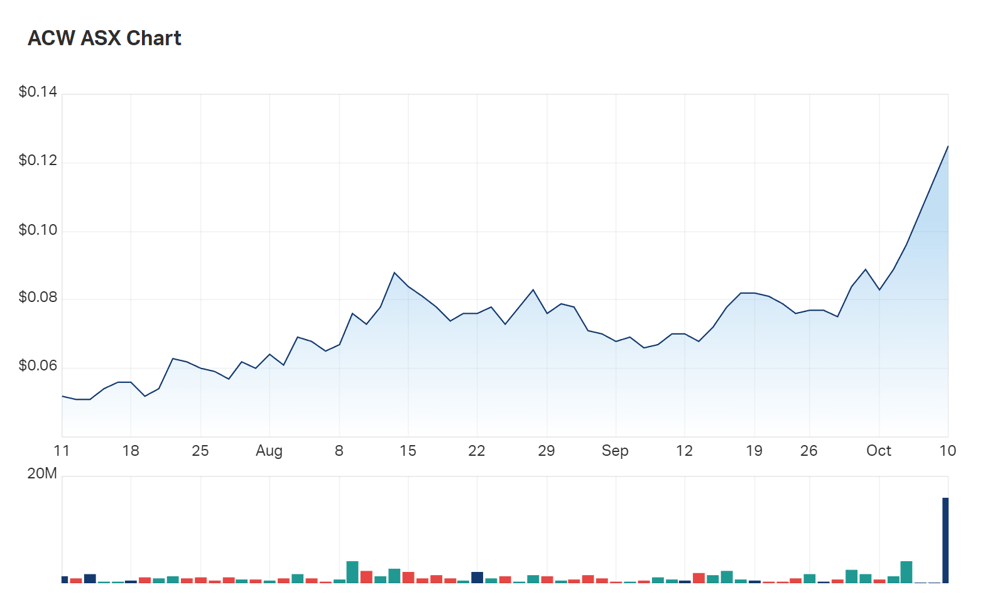
Actinogen (ASX:ACW) shareholders are among the lucky odd ones out surviving today’s red Monday for the ASX, with the company’s share price up 30% on the results of its Phase 2 Alzheimer’s Disease (AD) trials.
In 2014, Actionogen acquired the rights to an Alzheimer’s treatment product developed by the University of Edinburgh. That product was owned by a company called Corticrine, which Actinogen bought out.
Corticrine developed what is now called Xanamem by Actinogen, an oral tablet, as a cortisol disruptor, based on science inferring a link between cortisol and the onset of AD.
Actiongen’s Phase 2 trial results published today appear to confirm this association, with Xanamem successful in slowing the rate of progression of early AD.
The company claims it achieved a 60% reduction in the progression of disease in patients part of the latest trial.
Company moving on with next-stage trials
Actinogen CEO Dr. Steven Gourlay outlined the future potential of Xanamem in clinical contexts, based on today’s strong data.
“Xanamem has the potential to be a novel daily oral therapy for AD and other conditions,” Gourlay said.
“The results affirm our confidence in the upcoming clinical trials that will confirm if Xanamem can make a significant improvement in the lives of patients and their families.”
The company’s medical chief, Prof. Paul Rolan, echoed Gourlay’s comments, highlighting upcoming ‘next-stage’ trials.
“These results provide further validation of our AD program and reflect a significant step forward in the development of Xanamem as a new treatment for Alzheimer’s,” Rolan commented.
“Likely therapeutic effect”
Both company insiders are backed up by support from international AD expert Associate Prof. Michael Woodward, located at Australia’s Ramsay Health.
Worth noting is that Woodward’s comments focused more on Actinogen’s ability to diagnose AD, a secondary result of today’s data outside the larger highlight of a 60% reduction in onset speed.
“The positive datasets are encouraging and indicate a likely therapeutic effect of Xanamem in patients with early AD,” Woodward added.
“The use of blood levels to confirm the diagnosis of AD in future trials represents a practical and efficient method to select patients at risk of disease progression.”
Daily dose
Actinogen’s Xanamem is a 10mg oral tablet taken normally with water intended for daily use, should the company’s existing research be in order, and assuming FDA or TGA (Australia’s counterpart to the US regulator) approval down the line.
Not only does the drug appear to slow the progression of AD, but it also boosts cognitive quality, which today’s results also supported. Using an FDA-approved measurement metric for the neurodegenerative condition, management is hopeful it can best align itself with the US approvals process.
Dementia Australia predict in 2022, some 0.487m people live with Dementia, either early or severe in nature. By 2058, the group expects Alzheimer's to affect over 1m people.
There is a reason Actinogen is focused on the FDA.
The prevalence of AD in the US is significantly higher, at 6.5m US citizens with the condition at some stage of severity, according to the US Alzheimer’s Association.