Nasal spray hopeful Firebrick Pharma doubles valuation on ASX debut
Nasal spray business Firebrick hit highs of 65 cents from an IPO offer price of just 20 cents

Mentioned
KEY POINTS
- Firebrick hopes to complete its Phase 3 trials in 2022
- Plans to seek regulatory approval in Australia, the US and Europe
- Its patented Nasodine spray shows effectiveness against covid
Firebrick Pharma (ASX: FRE) became a two-bagger shortly after making its ASX debut at 12:30 pm AEDT.
The company’s stock hit intraday highs of 65 cents, from an IPO offer price of just 20 cents. Its currently trading at 53.5 cents.
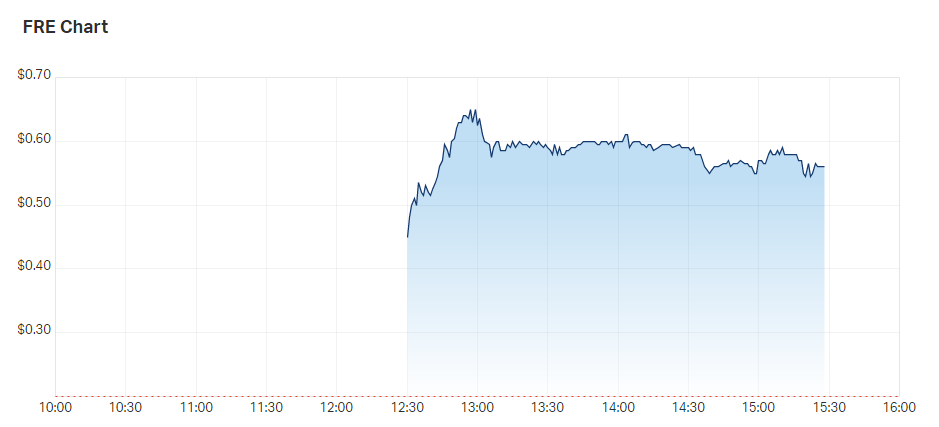
Firebrick intraday price chart
Firebrick is a small cap pharmaceutical hopeful with a breakthrough patent product called Nasodine Nasal Spray.
The nasal spray is designed to kill the germs that cause colds, targeting the nasal passages where the infection starts.
If approved by the Australian Therapeutic Goods Administration (TGA), Nasodine will be the first approved nasal spray that targets common colds and viruses from where they start, in the nose.
Firebrick’s IPO raised $7m, bringing its cash position to $13m. The company had an indicative market cap of $22.8m based on its offer price.
Upcoming catalysts
Firebrick Executive Chairman Dr Peter Molloy said “Nasodine is potentially only one successful Phase 3 clinical trial away from approval as treatment for the common cold in adults”.
“That Phase 3 trial is scheduled to take place in 2022. Once that trial is satisfactorily and successfully completed, we expect to be able to submit a dossier seeking regulatory approval of Nasodine in Australia and in the world’s two largest pharmaceutical markets, US and Europe.”
The trial to support regulatory approval is planned to commence in the Australian cold season (approximately March to September 2022).
Depending on overall recruitment success, the Firebrick said the trials could be "completed by the end of September 2022".
Nasodine vs. Covid
In 2020, Firebrick completed a laboratory study which confirmed its effectiveness against covid. The test showed that Nasodine killed 99.97% of SARS-CoV-2 in as little as 15 seconds.
This was followed by a pilot human trial, which showed that Nasodine reduced detectable infectious virus in the nasal passages in 5 out of 6 subjects.
Firebrick believes the nasal spray could play a part in reducing the viral shredding (transmissibility) from covid-positive people.
The Phase 2 study is planned to assess the impact of multiple Nasodine doses over a number of days. Firebrick intends to enlist more than 200 covid positive subjects for the study.

