Imugene to launch cancer medicine trial in Adelaide
Imugene (ASX:IMU) wins approval to launch Phase 2 stomach cancer trials in Adelaide
Source: Fisher & Paykel Healthcare
Mentioned
KEY POINTS
- Phase 2 Clinical Trial set for Queen Elizabeth Hospital in Adelaide focusing solely on stomach and upper stomach cancers
- Test will measure survival rates, changes to growth of stomach cancers, duration of response, and product safety
- Study to go ahead with full TGA approval after being shortlisted by US FDA late last year
Imugene (ASX:IMU) has won approval from the Australian Human Research Ethics Committee (HREC) to launch Phase 2 trials of its cancer immunotherapy product, HER-Vaxx.
Imugene's novel oncology immunotherapy product will be put through the testing process at Queen Elizabeth Hospital in Adelaide, and the company notes further clinical sites will be opened within Australia.
Imugene's HER-Vaxx is an injectable medicine designed to be used alongside chemotherapy treatments in patients with breast and or gastric cancer; the approval from HREC sees Imugene's Phase 2 clinical trial focusing specifically on gastric (stomach) cancer, and, gastroesophageal (upper stomach) cancer.

(Source: Cancer Australia) An Infographic detailing the incidence of stomach cancer in Australia through 2021
Imugene aiming for a cheaply-made, high-efficacy medicine
The vaccine product itself seeks to hamper the body's production of the HER-2 protein, which strong consensus in the medical community suggests enables cancerous cells to grow and spread in breast and gastric cancer patients.
By suppressing the creation of HER-2 proteins in the human body, Imugene seeks to commercialise a novel approach to immunotherapy in oncology settings.
Trial to create a baseline body of data in Australian context
Phase 2 trials will specifically examine patients who have not responded to cancer medication trastuzumab, and will be used alongside chemotherapy treatment pembrolizumab in the participating patient population.
The study ultimately seeks to set a benchmark for safety and response rate to HER-Vaxx in Australian patients.
The company will also measure duration of response, survival rates, and any changes in the speed of cancer growth.
HER-Vaxx was awarded Investigational New Drug (IND) status by the US Food and Drug Administration (FDA) late last year. The Australian Phase 2 trial will be conducted under the national Clinical Trials Notification (CTN) scheme and overseen by the Therapeutic Goods Administration (TGA).
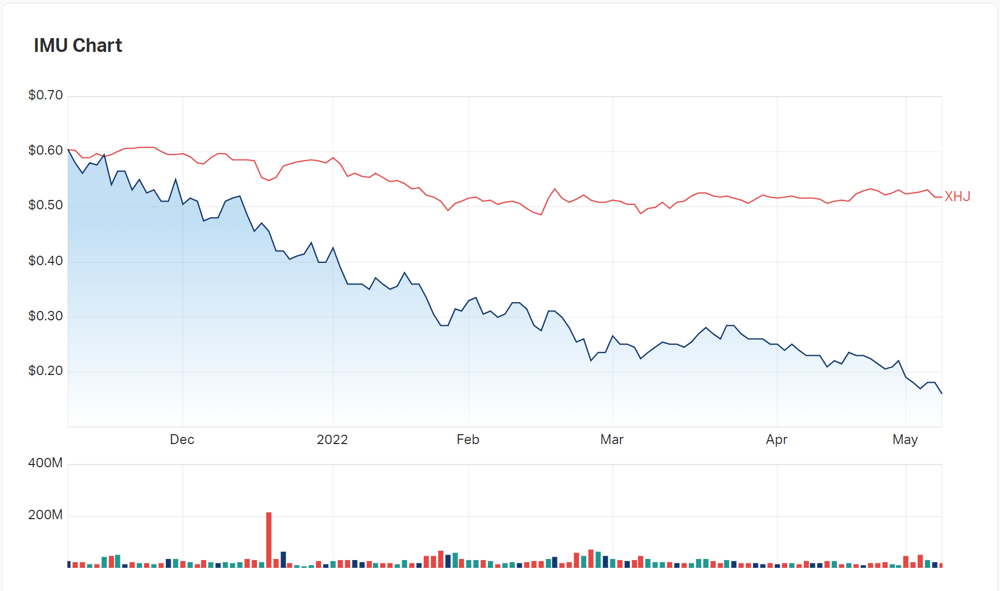
Value lost as healthcare sector takes a backseat
Despite making good headway this year for its commercial objectives, Imugene’s share price was down -11% on Monday afternoon and, year-to-date, the price has sunk -60% as the larger healthcare sector falls off the radar during a turbulent H1 CY22.
The price is currently trading at a significant discount to where it sat at the start of the year.