AnteoTech bounces back after posting quarterly business update
After reaching YTD lows yesterday, is AnteoTech set to recover from the dip?

KEY POINTS
- AnteoTech has posted a quarterly business update
- Most of the company's quarterly cash receipts came from tax refunds
- The company's focus has been gaining regulatory approval for its rapid antigen tests
AnteoTech (ASX: ADO) has recovered from recent losses to soar 12.82% today after posting a quarterly business update.
In the previous quarter, the company focussed on gaining regulatory approval for the EuGeni Reader and SARS-CoV-2 Ag Rapid Antigen Test (RAT).
AnteoTech also noted ongoing engagement with Australian industry and government bodies to lift the use and manufacturing of RATs. Outside of its Australian operations, the company progressed European sales and planning.
“Markets for rapid antigen testing (RATs) continued to be extremely dynamic during the December ‘21 quarter,” said CEO Derek Thomson in today’s letter to shareholders.
“In Australia, the laboratory-based PCR testing infrastructure buckled under the pressure of increased numbers of infections and Christmas travellers seeking to cross borders with required COVID-19 free test results.”
Energy
While AnteoTech is primarily considered to be in the healthcare sector, the company also has some exposure to the materials sector.
In today’s update, the company noted progress on the AnteoX (cross-linker additive) program. This program aims to increase electrode performance by improving binder chemistries for Lithium-ion battery electrodes.
Cash
Cash receipts for the quarter totalled $2,135,000. Most of this came from a cash refund of $1,965,463 under the Federal Government’s Research & Development (R&D) Tax Incentive Scheme.
Net cash outflows from operating activities was $0.611m.
As of 31 December, AnteoTech had $16.62 million cash on hand with zero debt.
Recent performance
It’s been a rocky start to the year for AnteoTech.
The company has been sliding for the past week, having fallen from a YTD high of $0.37 on 24 January to a YTD low of $0.195 yesterday.
Like many growth companies, the stock has been weighed by a shift towards higher interest rates.
Whether today’s bounce will fight this trend to continue upwards – marking yesterday’s lows as a trough – remains to be seen.
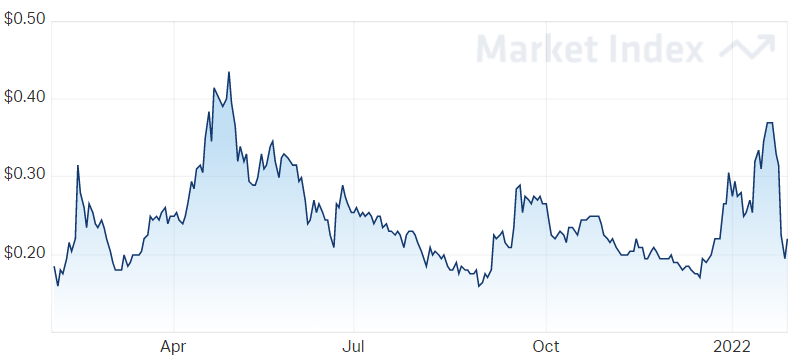
AnteoTech's price over the last year.
What do the brokers think?
Broker consensus is a strong sell.
However, it’s worth noting that the consensus was last updated on 6 January.