Ampila greenlighted to expand pancreatic cancer trial into Victoria
Ampila Therapeutics (ASX:ATX) wins second ethics approval to run pancreatic cancer trial

Source: iStock
Mentioned
KEY POINTS
- Human Research Ethics Committee hands Ampila Therapeutics approval to run simultaneous trial in NSW and now Vic
- Cancer Australia estimates pancreatic cancer is within the top three causes of cancer mortality in Australia
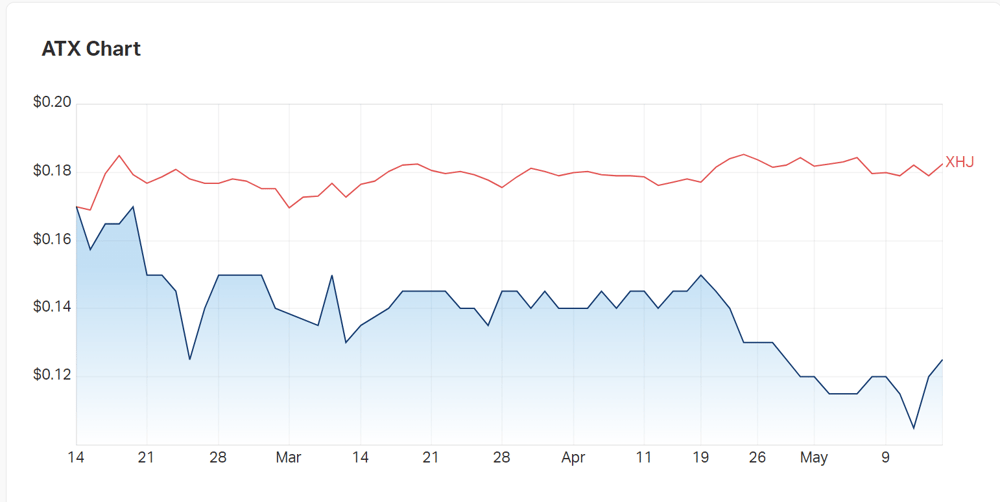
- Shares currently trading at a discount to price last year as sell-offs hit the healthcare sector
Ampila Therapeutics (ASX:ATX) is up 4% in pre-lunch trades as the company highlights its receipt of approvals from the Australian Human Research Ethics Committee (HREC) to conduct Phase 2 of its pancreatic cancer product trials.
The company's Focal Adhesion Kinase (FAK) inhibitor, called AMP945, seeks to stifle the body's production of enzymes which typically present in excessive amounts in Oncology patients.
FAK ultimately allows cancer cells in the body to spread and affects the ability of cells to 'stick' within the body.
Ampila is thus seeking to prove the efficacy of its novel cancer medication products by reducing the ability of cancer cells to spread in the human body. While Ampila looks at Oncology broadly, today's Phase 2 approvals from HREC relate specifically to pancreatic cancer patients.

(Source: Cancer Australia) Infographic showing pancreatic cancer stats in Australia for 2021
Simultaneous trials to run in NSW and Vic at once
Early last month, the company announced it had won ethics approval to conduct a trial in NSW; today's HREC approval allows the company to expand its trials into Victoria.
The company will now accelerate recruitment of patients into its trial. With all approvals needed under the company's belt, it is now free to focus wholly on the trial execution phase.
"This approval will allow us to recruit patients more rapidly and begin to generate results," Ampila chief Dr. John Lambert said.
"Our focus is now on execution of a high-quality trial."
Company not spared from sell-off trends
Looking at year to date performance, Ampila has lost 24% of its value as market-wide selloffs continue to slam the healthcare sector, though, the index has this week performed somewhat better than previous weeks gone by.
Year on year, the company is down 46%, but with trials underway, investor momentum may begin to pivot to a more positive outlook.
Cancer Australia highlighted in 2019 that pancreatic cancer is the fifth most common cause of cancer mortality in Australia; this was forecast to reach the third most common cause of cancer mortality in 2021.
1,600 men and 1,400 women died from pancreatic cancer in 2019.